Where does weight actually go?
#1
Token Canadian
Thread Starter
Join Date: Nov 2008
Location: Gagetown, New Brunswick
Posts: 1,555
Bikes: Cervelo S1, Norco Faze 1 SL, Surly Big Dummy, Moose Fatbike
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 200 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
Where does weight actually go?
With the discussions on bike weight, here's one on rider weight:
I have a question for those who understand the mechanics of nutrition.
Let's say we have someone who has some degree of body fat stores on their body. Let's also assume that they have been fasting for a little while so that their colon is clear of waste, their stomach and intestines are empty, as is their bladder.
This person then goes out on an epic ride and beasts themselves, burning a lot of energy. Let's say that the energy output is such that they "burn" 3500 calories of fat (which implies more than 3500 calories worth of work, because fat is not the only energy store tapped to produce work in the human body (I think 30% of total is the average during aerobic exercise but I'm fully willing to believe that I have that number wrong)
OK, so that fat isn't actually "burnt". It is processed in some sort of chemical reaction that breaks it down into something else, and in so doing, usable energy (in the chemical bonds of the fat) is released. But the actual matter composing the fat cannot be destroyed - it can be broken down, but the pre-reaction mass and post-reaction mass must be equal because we are talking chemical reactions, not nuclear.
So then, where does the mass of the "burnt" fat go? What is the mechanism for that mass actually leaving the body?
It has to be excreted somehow... so where does it go?
Put another way, using the same example, that person has "burnt" 3500 cal of fat which corresponds to a pound of fat. When would that weight loss actually be realized (ignoring weight lost through loss of fluid through perspiration and breathing)
DG
I have a question for those who understand the mechanics of nutrition.
Let's say we have someone who has some degree of body fat stores on their body. Let's also assume that they have been fasting for a little while so that their colon is clear of waste, their stomach and intestines are empty, as is their bladder.
This person then goes out on an epic ride and beasts themselves, burning a lot of energy. Let's say that the energy output is such that they "burn" 3500 calories of fat (which implies more than 3500 calories worth of work, because fat is not the only energy store tapped to produce work in the human body (I think 30% of total is the average during aerobic exercise but I'm fully willing to believe that I have that number wrong)
OK, so that fat isn't actually "burnt". It is processed in some sort of chemical reaction that breaks it down into something else, and in so doing, usable energy (in the chemical bonds of the fat) is released. But the actual matter composing the fat cannot be destroyed - it can be broken down, but the pre-reaction mass and post-reaction mass must be equal because we are talking chemical reactions, not nuclear.
So then, where does the mass of the "burnt" fat go? What is the mechanism for that mass actually leaving the body?
It has to be excreted somehow... so where does it go?
Put another way, using the same example, that person has "burnt" 3500 cal of fat which corresponds to a pound of fat. When would that weight loss actually be realized (ignoring weight lost through loss of fluid through perspiration and breathing)
DG
#3
Peloton Shelter Dog
With the discussions on bike weight, here's one on rider weight:
I have a question for those who understand the mechanics of nutrition.
Let's say we have someone who has some degree of body fat stores on their body. Let's also assume that they have been fasting for a little while so that their colon is clear of waste, their stomach and intestines are empty, as is their bladder.
This person then goes out on an epic ride and beasts themselves, burning a lot of energy. Let's say that the energy output is such that they "burn" 3500 calories of fat (which implies more than 3500 calories worth of work, because fat is not the only energy store tapped to produce work in the human body (I think 30% of total is the average during aerobic exercise but I'm fully willing to believe that I have that number wrong)
OK, so that fat isn't actually "burnt". It is processed in some sort of chemical reaction that breaks it down into something else, and in so doing, usable energy (in the chemical bonds of the fat) is released. But the actual matter composing the fat cannot be destroyed - it can be broken down, but the pre-reaction mass and post-reaction mass must be equal because we are talking chemical reactions, not nuclear.
So then, where does the mass of the "burnt" fat go? What is the mechanism for that mass actually leaving the body?
It has to be excreted somehow... so where does it go?
Put another way, using the same example, that person has "burnt" 3500 cal of fat which corresponds to a pound of fat. When would that weight loss actually be realized (ignoring weight lost through loss of fluid through perspiration and breathing)
DG
I have a question for those who understand the mechanics of nutrition.
Let's say we have someone who has some degree of body fat stores on their body. Let's also assume that they have been fasting for a little while so that their colon is clear of waste, their stomach and intestines are empty, as is their bladder.
This person then goes out on an epic ride and beasts themselves, burning a lot of energy. Let's say that the energy output is such that they "burn" 3500 calories of fat (which implies more than 3500 calories worth of work, because fat is not the only energy store tapped to produce work in the human body (I think 30% of total is the average during aerobic exercise but I'm fully willing to believe that I have that number wrong)
OK, so that fat isn't actually "burnt". It is processed in some sort of chemical reaction that breaks it down into something else, and in so doing, usable energy (in the chemical bonds of the fat) is released. But the actual matter composing the fat cannot be destroyed - it can be broken down, but the pre-reaction mass and post-reaction mass must be equal because we are talking chemical reactions, not nuclear.
So then, where does the mass of the "burnt" fat go? What is the mechanism for that mass actually leaving the body?
It has to be excreted somehow... so where does it go?
Put another way, using the same example, that person has "burnt" 3500 cal of fat which corresponds to a pound of fat. When would that weight loss actually be realized (ignoring weight lost through loss of fluid through perspiration and breathing)
DG
The ice cream bar in the freezer.
Go ahead.
#5
Banned
Join Date: Feb 2009
Location: Painville, USA
Posts: 1,914
Bikes: 2007 Tarmac Pro
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
Correct.
Of course, a tiny, tiny fraction of the mass is, of course, converted to the energy which is made available through the metabolism of the stores, but the amount is so small that only uber weight weenies would care.
Of course, a tiny, tiny fraction of the mass is, of course, converted to the energy which is made available through the metabolism of the stores, but the amount is so small that only uber weight weenies would care.
Last edited by Busta Quad; 05-23-09 at 05:13 PM.
#6
Senior Member
Join Date: Jun 2007
Location: A Coffin Called Earth. or Toronto, ON
Posts: 12,257
Bikes: Bianchi, Miyata, Dahon, Rossin
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 3 Post(s)
Likes: 0
Liked 5 Times
in
5 Posts
take an educational course in biology.
your liver and kidneys do most of the waste treatment work.
your liver and kidneys do most of the waste treatment work.
__________________
Food for thought: if you aren't dead by 2050, you and your entire family will be within a few years from starvation. Now that is a cruel gift to leave for your offspring. ;)
https://sanfrancisco.ibtimes.com/arti...ger-photos.htm
Food for thought: if you aren't dead by 2050, you and your entire family will be within a few years from starvation. Now that is a cruel gift to leave for your offspring. ;)
https://sanfrancisco.ibtimes.com/arti...ger-photos.htm
#7
Senior Member
Join Date: Nov 2006
Location: Houston, TX
Posts: 3,198
Bikes: 2007 Orbea Onix, 2007 Windsor The Hour, 2008 Kona Jake
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 1 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
#9
Senior Member
Join Date: Mar 2009
Posts: 179
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
I had a lot of gas after my first century. must have processed all those calories into methane.
does a 3500 kcal workout really take off a pound of fat?
does a 3500 kcal workout really take off a pound of fat?
#11
Senior Member
Join Date: Jan 2009
Posts: 628
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
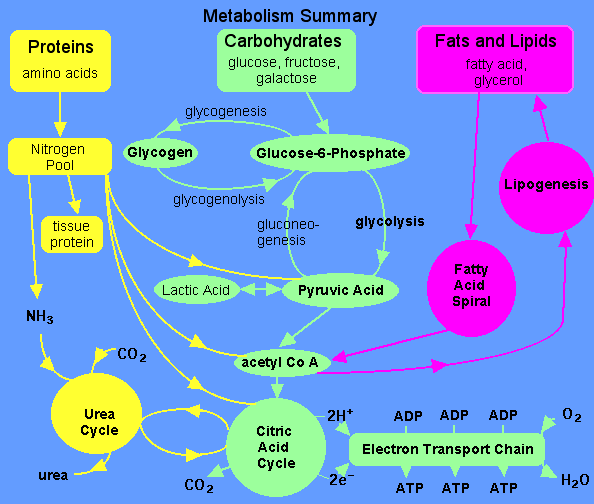
Memorize it and never ask the question again. (I had to remove 3 images)
Pyruvate Dehydrogenase & Krebs Cycle
 Pyruvate Dehydrogenase is a large complex containing many copies of each of three enzymes, E1, E2, and E3. The structure of the complex is depicted in figures on p. 769 & 774 of Biochemistry, 3rd Edition, by Voet & Voet.The inner core of the mammalian Pyruvate Dehydrogenase complex is an icosahedral structure consisting of 60 copies of E2.
Pyruvate Dehydrogenase is a large complex containing many copies of each of three enzymes, E1, E2, and E3. The structure of the complex is depicted in figures on p. 769 & 774 of Biochemistry, 3rd Edition, by Voet & Voet.The inner core of the mammalian Pyruvate Dehydrogenase complex is an icosahedral structure consisting of 60 copies of E2.
At the periphery of the complex are:
Enzyme
FAD accepts and donates 2 electrons with 2 protons (2 H):FAD + 2 e- + 2 H+ �� FADH2
 Thiamine pyrophosphate (TPP) is a derivative of thiamine (vitamin B1). Nutritional deficiency of thiamine leads to the disease beriberi. Beriberi affects especially the brain, because TPP is required for carbohydrate metabolism, and the brain depends on glucose metabolism for energy.
Thiamine pyrophosphate (TPP) is a derivative of thiamine (vitamin B1). Nutritional deficiency of thiamine leads to the disease beriberi. Beriberi affects especially the brain, because TPP is required for carbohydrate metabolism, and the brain depends on glucose metabolism for energy.

A proton readily dissociates from the C that is between N and S in the thiazole ring of TPP. The resulting carbanion (ylid) can attack the electron-deficient keto carbon of pyruvate. See also diagram p. 771.
Lipoamide includes a dithiol that undergoes oxidation and reduction. The carboxyl group at the end of lipoic acid's hydrocarbon chain forms an amide bond to the side-chain amino group of a lysine residue of E2.
A long flexible arm, including hydrocarbon chains of lipoate and the lysine R-group, links the dithiol of each lipoamide to one of two lipoate-binding domains of each E2. Lipoate-binding domains are themselves part of a flexible strand of E2 that extends out from thecoreof the complex.
The long flexible attachment allows lipoamide functional groups to swing back and forth between E2 active sites in the core of the complex and active sites of E1 & E3 in the outer shell of the complex.
The E3 binding protein (that binds E3 to E2) also has attached lipoamide that can exchange reducing equivalents with lipoamide on E2.
For diagrams showing the approximate arrangement of functional domains, based on structural studies of Pyruvate Dehydrogenase and a related enzyme see:
 Organic arsenicals are potent inhibitors of lipoamide-containing enzymes such as Pyruvate Dehydrogenase. These highly toxic compounds react with "vicinal" dithiols such as the functional group of lipoamide as shown at right.
Organic arsenicals are potent inhibitors of lipoamide-containing enzymes such as Pyruvate Dehydrogenase. These highly toxic compounds react with "vicinal" dithiols such as the functional group of lipoamide as shown at right.  In the overall reaction, the acetic acid generated is transferred to coenzyme A.
In the overall reaction, the acetic acid generated is transferred to coenzyme A.
The final electron acceptor is NAD+.
Complete structures of these coenzymes are presented in the section on bioenergetics.

 For example, acetyl CoA functions as:
For example, acetyl CoA functions as:
 Product inhibition by NADH and acetyl CoA: NADH competes with NAD+ for binding to E3. Acetyl CoA competes with Coenzyme A for binding to E2.
Product inhibition by NADH and acetyl CoA: NADH competes with NAD+ for binding to E3. Acetyl CoA competes with Coenzyme A for binding to E2.
Regulation by phosphorylation/dephosphorylation of E1: Specific regulatory Kinases and Phosphatases are associated with the Pyruvate Dehydrogenase complex within the mitochondrial matrix.
A Ca++-sensitive isoform of the phosphatase that removes phosphate residues from E1 is expressed in muscle cells. The increased cytosolic Ca++ that occurs during activation of muscle contraction can lead to Ca++ uptake by mitochondria. The higher Ca++ stimulates the phosphatase, and dephosphorylation activates Pyruvate Dehydrogenase. Thus mitochondrial metabolism may be stimulated during exercise.
Lecture notes relating to Krebs Cycle are not provided in the usual format, because lectures will be presented by students. Some questions on Krebs Cycle are included in the self-study quiz for this class.Select the interactive tutorial at right for information about the Krebs Citric Acid Cycle. Within the tutorial, drag the cursor over each enzyme name for information about that reaction.
Note that FADH2, listed as a product of succinate oxidation, is reoxidized to FAD as redox carriers within the Succinate Dehydrogenase complex pass electrons to coenzyme Q of the respiratory chain. Thus it would be more appropriate to list coenzyme QH2 as a product of the Succinate Dehydrogenase reaction. The initial acceptor, FAD, is included in the diagram for consistency with most textbooks.
Pyruvate Dehydrogenase & Krebs Cycle
Contents of this page:
Pathway localization
Pyruvate dehydrogenase
Roles of acetyl-coenzyme A
Regulation of pyruvate dehydrogenase
Krebs cycle
Pathway localization:
Glycolysis enzymes are located in the cytosol of cells. Pyruvate enters the mitochondrion to be metabolized further.
Pyruvate Dehydrogenase catalyzes oxidative decarboxylation of pyruvate, to form acetyl-CoA. The overall reaction is shown at right. Pathway localization
Pyruvate dehydrogenase
Roles of acetyl-coenzyme A
Regulation of pyruvate dehydrogenase
Krebs cycle
Pathway localization:
Glycolysis enzymes are located in the cytosol of cells. Pyruvate enters the mitochondrion to be metabolized further.
Mitochondrial compartments:
The mitochondrial matrix contains Pyruvate Dehydrogenase and enzymes of Krebs Cycle, plus other pathways such as fatty acid oxidation.
The mitochondrial outer membrane contains large VDAC channels, similar to bacterial porin channels, making the outer membrane leaky to ions and small molecules.
The inner membrane is the major permeability barrier of the mitochondrion. It contains various transport catalysts, including a carrier protein that allows pyruvate to enter the matrix. It is highly convoluted, with infoldings called cristae. Embedded in the inner membrane are constituents of the respiratory chain and ATP Synthase.

 Pyruvate Dehydrogenase is a large complex containing many copies of each of three enzymes, E1, E2, and E3. The structure of the complex is depicted in figures on p. 769 & 774 of Biochemistry, 3rd Edition, by Voet & Voet.The inner core of the mammalian Pyruvate Dehydrogenase complex is an icosahedral structure consisting of 60 copies of E2.
Pyruvate Dehydrogenase is a large complex containing many copies of each of three enzymes, E1, E2, and E3. The structure of the complex is depicted in figures on p. 769 & 774 of Biochemistry, 3rd Edition, by Voet & Voet.The inner core of the mammalian Pyruvate Dehydrogenase complex is an icosahedral structure consisting of 60 copies of E2.At the periphery of the complex are:
- 30 copies of E1 (itself a tetramer with subunits a2b2) and
- 12 copies of E3 (a homodimer), plus 12 copies of an E3 binding protein that links E3 to E2.
Enzyme
Abbreviated
Prosthetic Group Pyruvate Dehydrogenase E1
Thiamine pyrophosphate (TPP) Dihydrolipoyl Transacetylase E2
Lipoamide Dihydrolipoyl Dehydrogenase E3
FAD FAD (Flavin Adenine Dinucleotide) is a derivative of the B-vitamin riboflavin (dimethylisoalloxazine-ribitol). The flavin ring system undergoes oxidation/reduction as shown below. Whereas NAD+ is a coenzyme that reversibly binds to enzymes, FAD is a prosthetic group, that is permanently part of the complex. FAD accepts and donates 2 electrons with 2 protons (2 H):FAD + 2 e- + 2 H+ �� FADH2
 Thiamine pyrophosphate (TPP) is a derivative of thiamine (vitamin B1). Nutritional deficiency of thiamine leads to the disease beriberi. Beriberi affects especially the brain, because TPP is required for carbohydrate metabolism, and the brain depends on glucose metabolism for energy.
Thiamine pyrophosphate (TPP) is a derivative of thiamine (vitamin B1). Nutritional deficiency of thiamine leads to the disease beriberi. Beriberi affects especially the brain, because TPP is required for carbohydrate metabolism, and the brain depends on glucose metabolism for energy.
A proton readily dissociates from the C that is between N and S in the thiazole ring of TPP. The resulting carbanion (ylid) can attack the electron-deficient keto carbon of pyruvate. See also diagram p. 771.
A long flexible arm, including hydrocarbon chains of lipoate and the lysine R-group, links the dithiol of each lipoamide to one of two lipoate-binding domains of each E2. Lipoate-binding domains are themselves part of a flexible strand of E2 that extends out from thecoreof the complex.
The long flexible attachment allows lipoamide functional groups to swing back and forth between E2 active sites in the core of the complex and active sites of E1 & E3 in the outer shell of the complex.
The E3 binding protein (that binds E3 to E2) also has attached lipoamide that can exchange reducing equivalents with lipoamide on E2.
For diagrams showing the approximate arrangement of functional domains, based on structural studies of Pyruvate Dehydrogenase and a related enzyme see:
- a website of the laboratory of Wim Hol.
- an article by Milne et al. (Fig. 5, requires a subscription to J. Biol. Chem.).

 In the overall reaction, the acetic acid generated is transferred to coenzyme A.
In the overall reaction, the acetic acid generated is transferred to coenzyme A. The final electron acceptor is NAD+.
Complete structures of these coenzymes are presented in the section on bioenergetics.


The sequence of reactions catalyzed by the Pyruvate Dehydrogenase complex is summarized in Fig. 21-6 p. 770, and in the animation at right. The mechanism is depicted in greater detail on p. 771-772.
The reaction proceeds as follows:
The reaction proceeds as follows:
- The keto carbon of pyruvate reacts with the carbanion of TPP on E1 to yield an addition compound. The electron-pulling positively charged nitrogen of the thiazole ring promotes loss of CO2. What remains is hydroxyethyl-TPP.
- The hydroxyethyl carbanion on TPP of E1 reacts with the disulfide of lipoamide on E2. What was the keto carbon of pyruvate is oxidized to a carboxylic acid, as the disulfide of lipoamide is reduced to a dithiol. The acetate formed by oxidation of the hydroxyethyl moiety is linked to one of the thiols of the reduced lipoamide as a thioester (~).
- The acetate is transferred from the thiol of lipoamide to the thiol of coenzyme A, yielding acetyl CoA.
- The reduced lipoamide swings over to the E3 active site. Dihydrolipoamide is reoxidized to the disulfide, as 2 e- + 2 H+ are transferred to a disulfide on E3 (disulfide interchange).
- The dithiol on E3 is reoxidized as 2 e- + 2 H+ are transferred to FAD. The resulting FADH2 is reoxidized by electron transfer to NAD+, to yield NADH + H+.
 For example, acetyl CoA functions as:
For example, acetyl CoA functions as: - input to the Krebs Cycle, where the acetate moiety is further degraded to CO2.
- donor of acetate for synthesis of fatty acids, ketone bodies, and cholesterol.
 Product inhibition by NADH and acetyl CoA: NADH competes with NAD+ for binding to E3. Acetyl CoA competes with Coenzyme A for binding to E2.
Product inhibition by NADH and acetyl CoA: NADH competes with NAD+ for binding to E3. Acetyl CoA competes with Coenzyme A for binding to E2.Regulation by phosphorylation/dephosphorylation of E1: Specific regulatory Kinases and Phosphatases are associated with the Pyruvate Dehydrogenase complex within the mitochondrial matrix.
- Pyruvate Dehydrogenase Kinases catalyze phosphorylation of serine residues of E1, inhibiting the complex.
- Pyruvate Dehydrogenase Phosphatases reverse this inhibition.
A Ca++-sensitive isoform of the phosphatase that removes phosphate residues from E1 is expressed in muscle cells. The increased cytosolic Ca++ that occurs during activation of muscle contraction can lead to Ca++ uptake by mitochondria. The higher Ca++ stimulates the phosphatase, and dephosphorylation activates Pyruvate Dehydrogenase. Thus mitochondrial metabolism may be stimulated during exercise.
Lecture notes relating to Krebs Cycle are not provided in the usual format, because lectures will be presented by students. Some questions on Krebs Cycle are included in the self-study quiz for this class.Select the interactive tutorial at right for information about the Krebs Citric Acid Cycle. Within the tutorial, drag the cursor over each enzyme name for information about that reaction.
Note that FADH2, listed as a product of succinate oxidation, is reoxidized to FAD as redox carriers within the Succinate Dehydrogenase complex pass electrons to coenzyme Q of the respiratory chain. Thus it would be more appropriate to list coenzyme QH2 as a product of the Succinate Dehydrogenase reaction. The initial acceptor, FAD, is included in the diagram for consistency with most textbooks.
#12
Token Canadian
Thread Starter
Join Date: Nov 2008
Location: Gagetown, New Brunswick
Posts: 1,555
Bikes: Cervelo S1, Norco Faze 1 SL, Surly Big Dummy, Moose Fatbike
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 200 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
There we go...
So summarizing that... looks like C6H12O6 + 6 O2 -> 6 H2O + 6 CO2 + energy as a general mechanism, with the transport layer (ATP) constantly being broken down and rebuilt to make the process go.
So if cellular respiration is generating water as a byproduct, is that water always excreted directly, or does it form part of the general hydration "pool" (heh) meaning that it can depart as sweat, as water vapour in the breath, etc?
Not at all. Unlike biology (which I have never studied in depth) I have a very good handle on internal combustion. In fact, my car has special wide-range oxygen sensors in it, and has been programmed (by me) to combust very specific air/fuel ratios depending on the current RPM and engine load (as determined by the absolute manifold pressure) modified by factors for engine coolant temperature, ambient air temperature, and a few other factors (such as transient throttle tip-in enrichment)
If you like, I can calculate the amount of fuel flow at any given point of the engine's operation. And when I was racing and had tight control of the specific gravity of the fuel, I could predict the weight of fuel consumed during operation with high accuracy.
Biology is quite a bit more complicated; thus, the question.
DG
So summarizing that... looks like C6H12O6 + 6 O2 -> 6 H2O + 6 CO2 + energy as a general mechanism, with the transport layer (ATP) constantly being broken down and rebuilt to make the process go.
So if cellular respiration is generating water as a byproduct, is that water always excreted directly, or does it form part of the general hydration "pool" (heh) meaning that it can depart as sweat, as water vapour in the breath, etc?
you must wonder where all that gasoline you put in your car goes to as well.
If you like, I can calculate the amount of fuel flow at any given point of the engine's operation. And when I was racing and had tight control of the specific gravity of the fuel, I could predict the weight of fuel consumed during operation with high accuracy.
Biology is quite a bit more complicated; thus, the question.
DG
#13
Senior Member
Join Date: Jan 2009
Posts: 628
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
There we go...
So summarizing that... looks like C6H12O6 + 6 O2 -> 6 H2O + 6 CO2 + energy as a general mechanism, with the transport layer (ATP) constantly being broken down and rebuilt to make the process go.
So if cellular respiration is generating water as a byproduct, is that water always excreted directly, or does it form part of the general hydration "pool" (heh) meaning that it can depart as sweat, as water vapour in the breath, etc?
So summarizing that... looks like C6H12O6 + 6 O2 -> 6 H2O + 6 CO2 + energy as a general mechanism, with the transport layer (ATP) constantly being broken down and rebuilt to make the process go.
So if cellular respiration is generating water as a byproduct, is that water always excreted directly, or does it form part of the general hydration "pool" (heh) meaning that it can depart as sweat, as water vapour in the breath, etc?
H20 loss from Respiration would come from the breath. Weight loss from sweat is a whole separate mechanism that is fighting the heat generated from respiration. Weight loss from respiration would need more calories taken in and turned to fat/muscle. Weight loss from sweat only takes a glass of water (0 calories) to be replenished.
#16
Senior Member
Join Date: May 2006
Posts: 427
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
Great responses... from the last graph, you can see that fatty acids (stored as triglycerides in fat cells) will be degenerated in their own pathway to acetyl Co-A, where they will enter the krebs cycle. Thus the molecules you ingested as a burger last year end up going out the same way as the sugar in the gatorade you just drank.
The phrase "sweating away the pounds" pretty much accurately sums it up.
J
Biologist, Cyclist
The phrase "sweating away the pounds" pretty much accurately sums it up.
J
Biologist, Cyclist
__________________
"I love fools� experiments. I am always making them." - Darwin
"I love fools� experiments. I am always making them." - Darwin
Last edited by austinspinner; 05-23-09 at 08:45 PM.
#18
Senior Member
Join Date: Jan 2009
Posts: 628
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
Great responses... from the last graph, you can see that fatty acids (stored as triglycerides in fat cells) will be degenerated in their own pathway to acetyl Co-A, where they will enter the krebs cycle. Thus the molecules you ingested as a burger last year end up going out the same way as the sugar in the gatorade you just drank.
The phrase "sweating away the pounds" pretty much accurately sums it up.
J
Biologist, Cyclist
The phrase "sweating away the pounds" pretty much accurately sums it up.
J
Biologist, Cyclist
However if you are sweating because you are working out (as opposed to sitting in a sauna), you can have a more permanent weight loss.
Semantics? Maybe.
#19
Senior Member
Join Date: Nov 2006
Location: Los Alamos, NM
Posts: 903
Bikes: 2008 fetish illustre
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 10 Post(s)
Likes: 0
Liked 8 Times
in
1 Post
^^ +1 to the cat WTF pic!
My understanding simply stops at
[energy expended] = [energy consumed]
Slightly on-topic question though: I've heard once you create a fat cell, that it never really goes away. Sure, you deplete it, but the cell itself stays around, just waiting to fill up again. Same thing I've heard about muscle cells. Bunk?
My understanding simply stops at
[energy expended] = [energy consumed]
Slightly on-topic question though: I've heard once you create a fat cell, that it never really goes away. Sure, you deplete it, but the cell itself stays around, just waiting to fill up again. Same thing I've heard about muscle cells. Bunk?
#20
Senior Member
Join Date: Jan 2009
Posts: 628
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
That is the theory on "fat cells' you gain them up to a certain age then you always have them (barring liposuction) and as you say they just fill and empty. Not sure on muscle cells since if you go into starvation your body can actually consume your muscle.
#22
Senior Member
Join Date: May 2006
Posts: 427
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
Semantics perhaps. Sauna sweat = H2O out. Exercise + C, H, O in fat cells = C, H, O in CO2 exhaled +H2O sweated out. Rehydration = H2O in, no new fat.
__________________
"I love fools� experiments. I am always making them." - Darwin
"I love fools� experiments. I am always making them." - Darwin
#23
Senior Member
Join Date: Feb 2009
Location: K.F., Orygun
Posts: 905
Bikes: 08 Giant Boulder, 08 Scattante XLR
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 1 Time
in
1 Post
That's all wrong anyway - we all have unborn twins in a cross-dimensional karmic system of balancing connections. If I ride my ass off and keep it at 170, there's a fat guy in a parallel world wondering why the heck he can't shed a pound.
#24
Ride to Ride
Join Date: Jun 2007
Location: Cary, NC
Posts: 641
Bikes: Fuji C7 full DA
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
#25
Ride to Ride
Join Date: Jun 2007
Location: Cary, NC
Posts: 641
Bikes: Fuji C7 full DA
Mentioned: 0 Post(s)
Tagged: 0 Thread(s)
Quoted: 0 Post(s)
Likes: 0
Liked 0 Times
in
0 Posts
... still missing the icecream connection, though I'm guessing it has something to do with a thermodynamic offset?