Originally Posted by
masi61
I think the OP may have been asking if anyone would do an extended (self contained) tour with a waxed chain system. I took this to mean that you would bring a pre-waxed back up chain and also a pan with solidified wax with a tight fitting lid that could be used on a backpacking cook stove. As impractical as some might think this sounds, I like the idea. I have enjoyed waxing my 2 road bike chains the past 4 years and I keep getting more little tips from places like Molten Speed Wax & Silca on how to do it better. For example: on a Silca video, Josh suggested pre-cleaning a spent waxed chain with boiling water before plunging it into melted wax. I have been doing this immediately after rain rides & when having the melted wax standing by - you plunge the wet (but rinsed of grit with boiling water) chain into the molten wax & you can observe the water bubbling off right away. You slosh back and forth then flip & pull from the wax pot when the bubbling stops. This works great for rain riding & I see no reason why this method could not be taken on a long distance tour. If anybody had an idea for a sealed small pan that would hold half a pound of Silca wax with a snug sealed lid - do share!
There is so much horse crap being spread by the makers and sellers of wax products that I�m surprised they don�t have to be followed around by a cart with a broom and shovel.

Where do they get their ideas? First, why remove wax with water? Why introduce water into the equation? Hot wax is a nice solvent for cold wax. It just melts. No need for washing the wax off with hot water because hot water doesn�t actually wash off wax. If the wet chain is then dropped into a hot wax, the water doesn�t move off into the wax either. It becomes encapsulated against the metal and just sits there, corroding the metal underneath. There are many bizarre and dumb cleaning methods that have little rhyme or reason for existing.
Below is a post from another discussion which was about oil but it also applies to wax and the effect of water on that wax.
Originally Posted by
cyccommute
I found
a paper in Science Direct from someone in the petroleum business. The article is mostly about water that is entrained in petroleum flowing in a pipeline but it is still applicable to a nearly stagnant example on a bike chain. As seen below, they present a very good graphic as to what happens when water comes in contact with an oil wetted surface. I�ll put this in context of chain that is being wetted due to rain or splashing. In no. 1, the water rolls along on top of the wetted surface. Eventually�either through gravity, mixing, electrostatic seeking of the metal by the water, or a combination of all three�the water will penetrate the oil (no.2). Yes, it will be a sphere with a high angle because the water is trying to reduce the surface interaction with the oil as much as possible. At number 3 the water will �pin� or spread out under the edges of the oil to make contact with the metal. It will continue to spread and thin as the water is now trying to make as much contact with the metal with which it is more electrostatically compatible and will try to maximize that interaction. It will lift the oil off the surface and in addition to making contact with the metal start oxidation reactions.
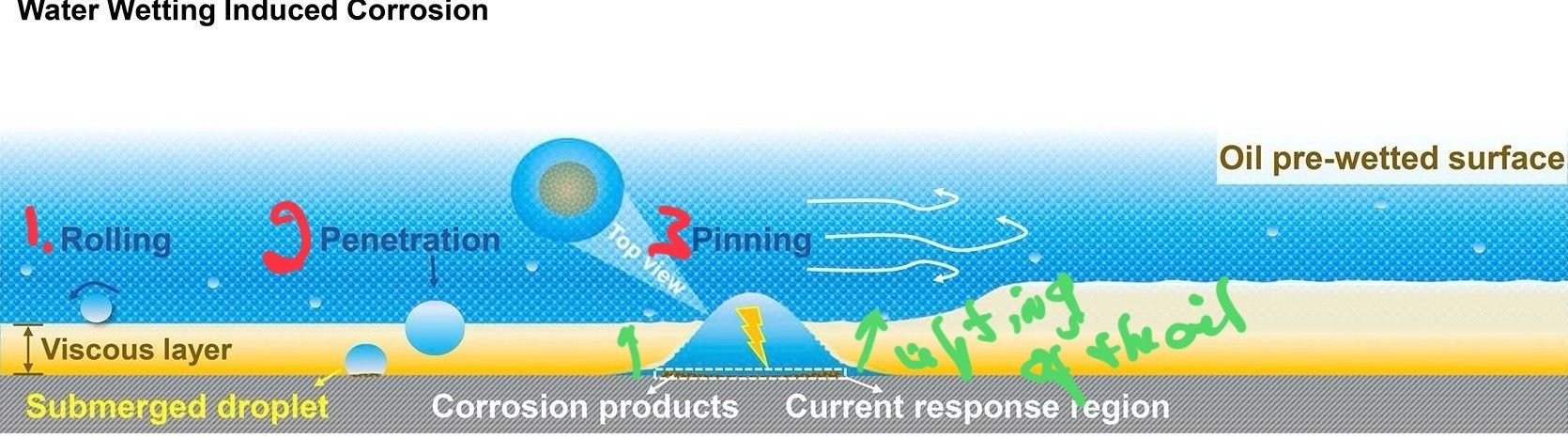
The lower half of the illustration shows what happens when oil is placed on top of a surface that is water wetted. The oil (bicycle oil, WD-40, PB Blaster, etc) rides on top of the oil, trapping the water against the surface where it can go about its merry way and corrode the metal. Add in some salt from sea water and the corrosion rate is going to be increased significantly. Rinsing the chain with denatured alcohol or acetone after a water wash will do little to remove the oil�they are both nearly as polar as water�but it will remove any water that hasn�t had a chance to penetrate into the oil layer and will probably remove any water that
has penetrated if it is still sticking above the oil.

Bottom line, hot wax itself is sufficient for removing cold wax.